Glossary of Terms used for Water and Watershed Management in Alberta
Lake Specific Glossary (from the Lakekeeper’s Manual – © BC Lake Stewardship Society
A B C D E F H K L M N O P R S T W Z
Acid
Corrosive substances with a pH of less than 7.0; acidity is caused by high concentrations of hydrogen ions.8
Aerobic
In the presence of, or requiring, oxygen.1
Algae
A plant or plantlike organism of any of several classes of chiefly aquatic, usually chlorophyll-containing, nonvascular organisms that usually include the green, yellow-green, brown, and red algae and the blue-green algae (also known as cyano bacteria).7
Algal Bloom
Population explosion of algae in surface waters due to an increase in plant nutrients such as nitrates and phosphates.8 Usually due to excessive blue green algae growth.
Alkalinity
(aka Basic) The ability of water, or other substances, to absorb high concentrations of hydrogen ions. Substances with a pH greater than 7.0 are considered alkaline. A measure of the amount of carbonates, bicarbonates, and hydroxide present in water. Low alkalinity is the main indicator of susceptibility to acid rain. Increasing alkalinity is often related to increased algae productivity. Expressed as milligrams per litre (mg/L) of calcium carbonate (CaCO3), or as microequivalents per litre (?eq/L). 20 ?eq/L = 1 mg/L of CaCO3.8
Amictic Lake
Lake with the absence of circulation periods, as in permanently ice-covered lakes.3
Ammonia
A form of nitrogen found in organic materials and many fertilizers. It is the first form of nitrogen released when organic matter decays. It can be used by most aquatic plants and is therefore an important nutrient. It converts rapidly to nitrate (NO3 ) if oxygen is present. The conversion rate is related to water temperature. Ammonia is toxic to fish at relatively low concentrations in pH-neutral or alkaline water. Under acid conditions, non-toxic ammonium ions (NH4+) form, but at high pH values the toxic ammonium hydroxide (NH4OH) occurs. The water quality standard for fish and aquatic life is 0.02 mg/L of NH4OH.8
Ammonification
Production of ammonia (NH3) from organic nitrogenous compounds by decay of dead material and metabolism in living organisms.3
Anaerobic
Relating to a process that occurs with little or no oxygen present.1
Anoxic
Lack of oxygen. An adjective usually used to describe a microbial habitat.1
Bacteria
Tiny, unicellular organisms that reproduce by cell division and usually have cell walls; can be shaped like spheres, rods or spirals and can be found in virtually any environment.1
Benthic
Referring to bottom zones or bottom-dwelling forms.3
Benthos
Animals and plants living on or within the substrate of a water body (freshwater, estuarine or marine).1
Biodiversity
The existence of a wide range of different types of organisms in a given place at a given time.1
Blue Listed
Includes any indigenous species or subspecies (taxa) considered to be vulnerable in British Columbia. Vulnerable taxa are of special concern because of characteristics that make them particularly sensitive to human activities or natural events. Blue-listed taxa are at risk, but are not extirpated, endangered or threatened. 4
Carnivores
An organism that eats animals.1
Chlorophyll
A green, light-absorbing pigment found in plants and other photosynthetic organisms. A magnesium-porphyrin complex, it is an essential electron donor in photosynthesis.1 The amount of chlorophyll present in lake water depends on the amount of algae and is therefore used as a common indicator of water quality. 8
Clarity
A Secchi disk is a 20 cm (8 inch) diameter plate with alternating quadrants painted black and white that is used to measure water clarity (light penetration). The disc is lowered into water until it disappears from view. It is then raised until just visible. An average of the two depths, taken from the shaded side of the boat, is recorded as the Secchi disk reading. For best results, the readings should be taken on sunny, calm days, without wearing sunglasses.8
Cold Monomictic Lake
Cold monomictic lakes have a water temperature that never exceeds 4(C and a period of circulation in the summer. These lakes are mainly found in the Arctic mountains and usually have some contact with glaciers or permafrost.
Conductivity
Measures a water’s ability to conduct an electric current. Conductivity is reported in micromhos per centimeter (?mhos/cm) and is directly related to the total dissolved inorganic chemicals in the water. Values are commonly two times the water hardness unless the water is receiving high concentrations of contaminants introduced by humans.8
Consumers
A nutritional grouping in the food chain of an ecosystem, composed of heterotrophic organisms, chiefly animals, which ingest other organisms or particulate organic matter.2
Decomposers
Heterotrophic organisms (including bacteria and fungi) which break down the complex compounds of dead organisms, absorbs some decomposition products, and releases substances usable by consumers.2
Decomposition
The breakdown of dead organic material.
Detritus
Undissolved organic or inorganic matter resulting from the decomposition of parent material.1
Dimictic Lake
Lake with two circulation or mixing periods, (spring and autumn), per year; dimictic lakes circulate after the ice melts before summer stratification and after temperature stratification is destroyed and before ice forms.3 The common lake of interior BC.
Dissolved Oxygen
The amount of free oxygen absorbed by the water and available to aquatic organisms for respiration; amount of oxygen dissolved in a certain amount of water at a particular temperature and pressure, often expressed as a concentration in parts of oxygen per million parts of water (ppm).8
Epilimnion
The upper, well-mixed, well-illuminated, nearly isothermal region of a typical stratified lake.3
Erosion
Movement of soil by water or wind.8
Eutrophic
Rich in dissolved nutrients, photosynthetically productive and often deficient in oxygen during warm weather.1
Eutrophication
The process by which lakes and streams are enriched by nutrients, and the resulting increase in plant and algae growth. This process includes physical, chemical, and biological changes that take place after a lake receives inputs for plant nutrients–mostly nitrates and phosphates–from natural erosion and runoff from the surrounding land basin. The extent to which this process has occurred is reflected in a lake’s trophic classification: oligotrophic (nutrient poor), mesotrophic (moderately productive), and eutrophic (very productive and fertile).8
Evapotranspiration
Loss of water by evaporation from the soil and transpiration from plants.1
Exotic Species
Plant or animal species introduced into an area where they do not occur naturally; non-native species.1 Examples area Eurasian Milfoil and Purple Loosestrife.
Extirpated
Taxa that no longer exist in the wild in Alberta, but do occur elsewhere.4
Flushing rate
Flushing rate is the rate of water replacement in a lake. It’s unit of measure is times/year. Conversely, retention time is the average length of time water resides in a lake, ranging from several days in small impoundments to many years in large seepage lakes. Retention time is important in determining the impact of nutrient inputs. Long retention times result in recycling and greater nutrient retention in most lakes. Calculate retention time by dividing the lake volume by the volume of water passing through the lake in on year.
Food chain
The transfer of food energy from plants through herbivores to carnivores. An example: insect-fish-bear or the sequence of algae being eaten by small aquatic animals (zooplankton) which in turn are eaten by small fish which are eaten by larger fish and eventually by people or predators.8
Herbivores
An animal that consumes herbaceous vegetation.1
Herbaceous
Refers to a plant that has a non-woody stem and which dies back at the end of the growing season.1
Hydrological Cycle
Pertaining to the cyclic phenomena of waters of the earth – successively as condensation, precipitation, runoff, storage and evapotransporation, and quantitatively as to distribution and concentration.
Hypolimnion
Lowermost, noncirculating layer of cold water in a typical, thermally stratified lake, usually deficient in oxygen.1
Kjeldhal Nitrogen
The most common analysis run to determine the amount of organic nitrogen in water. The test includes ammonium and organic nitrogen.8
Lake Order
A whole number ranking assigned to lakes based on their distance from headwater sources, by number o contributing streams. With low numbered lakes closer to sources such as glacier runoffs and springs, and higher order lakes further away from these sources, with higher numbers of contributing streams and rivers. Related to stream order.
Limnetic
Of, pertaining to, or inhabiting the pelagic region of a body of fresh water.2
Littoral
Pertaining to or along the shore, particularly to describe currents, deposits, and drift.1
Macrophytes
A member of the rooted aquatic plant life of an area, especially of a body of water.1
Mesomictic Lake
Lake with partial circulation, the lower denser layers never mixing with the upper.3
Metalimnion
The central stratum between the epilimnion and hypolimnion in a stratified lake; the region occupied by the thermocline.3
Morphometry
Measurement of external form.7 Lake morphometry includes maximum and average depth, surface area, volume, shoreline length,etc.
Nitrate
An inorganic form of nitrogen important for plant growth. Nitrogen is in this stable form when oxygen is present. Nitrate often contaminates groundwater when water originates from manure pits, fertilized fields, lawns or septic systems. High levels of nitrate-nitrogen (over 10 mg/L) are dangerous to infants and expectant mothers. A concentration of nitrate-nitrogen (NO3-N) plus ammonium-nitrogen (NH4-N) of 0.3 mg/L in spring will support summer algae blooms if enough phosphorus is present.8
Nitrification
The conversion of ammonia (NH3) to nitrate (NO3-).1
Nitrite
A form of nitrogen that rapidly converts to nitrate (NO3-) and is usually included in the NO3- analysis.8
Nitrogen Fixation
The conversion of atmospheric nitrogen (N2) into an organic form usable by plants and other organisms; nitrogen is typically fixed by bacteria that live in nodules on the roots of legumes and similar plants.1
Nutrients
Elements or substances such as nitrogen and phosphorus that are necessary for plant growth. Large amounts of these substances can become a nuisance by promoting excessive aquatic plant growth.8
Oligomictic Lake
Oligomictic lakes are characterised by a rare circulation at irregular intervals and have a temperature that is always above 4(C. This type of lake is found in the tropics.
Oligotrophic
Describes a body of water in which nutrients are in low supply.1
Orthophosphorus
Dissolved inorganic phosphorus.3 The dissolved inorganic form of phosphorus that is immediately bio-available for absorption by algae. Also can be referred to as soluble reactive phosphorus (SRP).
Pathogen
A disease-producing agent; usually applied to a living organism. Generally, any viruses, bacteria, protozoans or fungi that cause disease. 5
Pelagic
The main open water portion of a lake, beyond the extent of rooted vegetation and above the lake’s profundal zone
Periphyton
Algae and associated microorganisms growing attached to any submerged surface, such as rocks or plants.1
Phosphorus
Key nutrient influencing plant growth. Soluble reactive phosphorus (orthophosphorus) is the amount of phosphorus in solution that is available to plants. Total phosphorus includes the amount of phosphorus in solution (reactive) and in particulate form.8
Photosynthesis
Process through which light energy, water, and carbon dioxide are converted to carbohydrate and oxygen in the presence of chlorophyll. Occurs in plants, algae, cyanobacteria and lichens.1
Phytoplankton
Microscopic plants found in the water. Algae or one-celled (phytoplankton) or multicellular plants either suspended in water (plankton) or attached to rocks and other substrates (periphyton). Their abundance, as measured by the amount of chlorophyll a (green pigment) in an open water sample, is commonly used to classify the trophic status of a lake. Numerous species occur. Algae are an essential part of the lake ecosystem and provides the food base for most lake organisms, including fish. Phytoplankton populations vary widely from day to day, as life cycles are short.8
Plankton
Small plant organisms (phytoplankton and nanoplankton) and animal organisms (zooplankton) that float or swim weakly though the water.8
Polymictic Lake
Lake with many circulation or mixing periods per year or nearly continuous ciculation.3
ppm
parts per million; units per equivalent million units; equal to milligrams per litre (mg/L).8
Precipitate
A solid material which forms and settles out of water as a result of certain negative ions (anions) combining with positive ions (cations).8 Metals tend to precipitate in the presence of oxygen.
Producer
An organism that uses light to synthesize new organic material from carbon dioxide.1
Profundal
The region occurring below the limnetic (pelagic) zone. The main sediment zone of a lake, the non-vegetated lake bottom sediment.
Red Listed
Includes any indigenous species or subspecies (taxa) considered to be extirpated, endangered or threatened in British Columbia. Extirpated taxa no longer exist in the wild in British Columbia, but do occur elsewhere. Endangered taxa are facing imminent extirpation or extinction. Threatened taxa are likely to become endangered if limiting factors are not reversed. Red-listed taxa include those that have been, or are being, evaluated for these designations. 4
Respiration
Complex process that occurs in the cells of plants and animals in which nutrient organic molecules such as glucose combine with oxygen and produce carbon dioxide, water, and energy. It is the reverse reaction of photosynthesis. Respiration consumes oxygen (02) and releases carbon dioxide (CO2). It also takes place as organic matter decays.8
Restoration
Measures undertaken to return a degraded ecosystem’s functions and values, including its hydrology, plant and animal communities, and/or portions thereof, to a less degraded ecological condition.1
Secchi Disk
An 20 cm (8 inch) diameter plate with alternating quadrants painted black and white that is used to measure water clarity (light penetration). The disc is lowered into water until it disappears from view. It is then raised until just visible. An average of the two depths, taken from the shaded side of the boat, is recorded as the Secchi disc reading. For best results, the readings should be taken on sunny, calm days.8
Sedimentation
The process of or accumulation of sand and dirt settling on the bottom of a lake.
Specific Conductance
The removal, transport, and deposition of detached soil particles by flowing water or wind. Accumulated organic and inorganic matter on the lake bottom. Sediment includes decaying algae and weeds, marl, and soil and organic matter eroded from the lake’s watershed.8 See Conductivity
Stewardship
The conducting, supervising, or managing of something; especially: the careful and responsible management of something entrusted to one’s care (stewardship of our natural resources).7
Stratification
The layering of water due to differences in density. Water’s greatest density occurs at 4oC (39oF ). As water warms during the summer, it remains near the surface while colder water remains near the bottom. Wind mixing determines the thickness of the warm surface water layer (epilimnion), which usually extends to a depth of about 6.5 m (20 feet). The narrow transition zone between the epilimnion and cold bottom water (hypolimnion) is called the metalimnion or thermocline.8
Suspended Solids
A measure of the particulate matter in a water sample, expressed in milligrams per liter. When measured on inflowing streams, it can be used to estimate the sedimentation rate of lakes or impoundments.8
Thermocline
Layer of water between the warmer surface zone and the colder deep zone of a thermally stratified body of water. In the thermocline, temperature decreases rapidly with depth.1
Transpiration
The passage of water in plants from the roots through the vascular system to the stoma of the leaves and into the atmosphere.1
Trophic Levels
A classification of organisms according to what they eat.8
Trophic State
Eutrophication is the process by which lakes are enriched with nutrients, increasing the production of rooted aquatic plants and algae. The extent to which this process has occurred is reflected in a lake’s trophic classification or state: oligotrophic (nutrient poor), mesotrophic (moderately productive), and eutrophic (very productive and fertile).8
Turbidity
Degree to which light is blocked because water is muddy or cloudy.8
Turnover
(or Overturn) Fall cooling and spring warming of surface water changes it’s density, and gradually makes temperature and density uniform from top to bottom. This allows wind and wave action to mix the entire lake. Mixing allows bottom waters to contact the atmosphere, raising the water’s oxygen content. However, warming may occur too rapidly in the spring for mixing to be effective, especially in small sheltered kettle lakes.8
Warm Monomictic Lake
Ice-free lake with one period of complete circulation during the cold time of year; temperature stratification occurs in summer.3
Water Quality
A term used to describe the chemical, physical, and biological characteristics of water, usually in respect to its suitability for a particular purpose.5
Watershed
The total area above a given point on a watercourse that contributes water to its flow; the entire region drained by a waterway or watercourse that drains into a lake or reservoir.1
Zooplankton
A community of floating, aquatic, minute animals and non-photosynthetic protists.1
The definitions listed above have been adapted from the following sources:
1. Biotech Life Science Dictionary. 1998. Indiana Institute for Molecular and Cellular Biology. 25 Mar. 2002.
2. Parker, Sybil, ed. Dictionary of Bioscience. New York: McGraw-Hill, 1997.
3. Cole, G.A. Textbook of Limnology 4th Ed. (Glossary). Illinois: Waveland Press Inc, 1994.
4. British Columbia Conservation Data Centre. 2001. Ministry of Sustainable Resource Management. 25 Mar. 2002.
5. Water Science Glossary of Terms. 2002. Geophysics Istanbul University. 25 Mar. 2002.
6. Glossary of Natural History Terms: Volume #5 (Ecological Terms). 2002. University of California Museum of Paleontology. 25 Mar. 2002.
7. Collegiate Dictionary. 2002. Merriam Webster Incorporated. 25 Mar. 2002.
8. Glossary of Lake and Water Terms. 1998. Wisconsin Department of Natural Resources. 25 Mar. 2002.


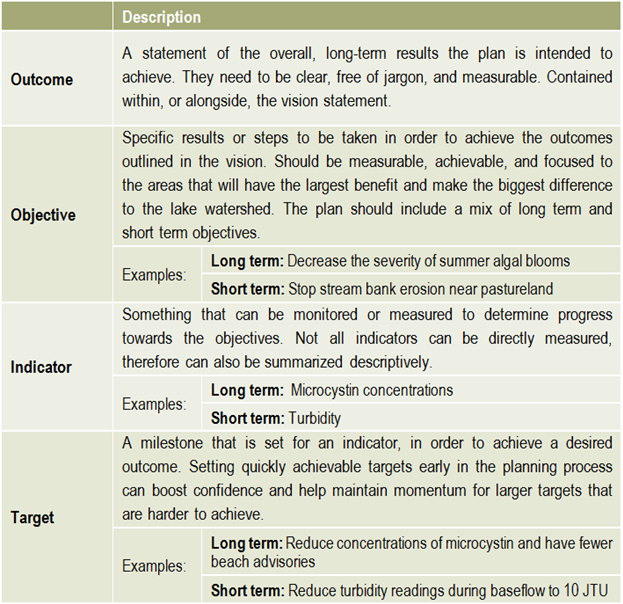
 The members of the steering committee will continue to play a strong role in facilitating and tracking implementation actions. This includes any actions they were responsible for, as well as tracking other committees and sector’s actions and progress made towards achieving the plan’s outcomes. Ongoing communication is essential to successful implementation and achieving outcomes, therefore a regular reporting mechanism could be set up in order to provide regular evaluation of the plan.
The members of the steering committee will continue to play a strong role in facilitating and tracking implementation actions. This includes any actions they were responsible for, as well as tracking other committees and sector’s actions and progress made towards achieving the plan’s outcomes. Ongoing communication is essential to successful implementation and achieving outcomes, therefore a regular reporting mechanism could be set up in order to provide regular evaluation of the plan.

 Reporting is an essential component of any watershed management planning and implementation process. There are two main types of reporting that should be shared with stakeholders on a regular basis: implementation reporting & effectiveness reporting.
Reporting is an essential component of any watershed management planning and implementation process. There are two main types of reporting that should be shared with stakeholders on a regular basis: implementation reporting & effectiveness reporting.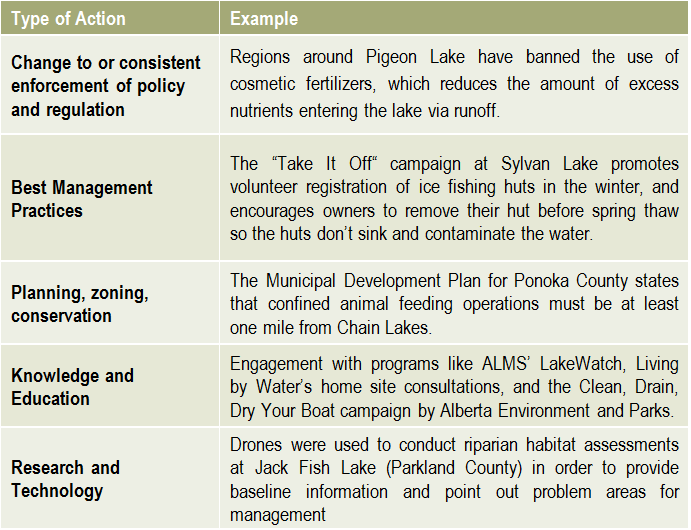 There is no limit to the number or types of lake management actions, but they typically fall into the categories on the right.
There is no limit to the number or types of lake management actions, but they typically fall into the categories on the right.

 Helpful resources
Helpful resources The development of a lake watershed management plan provides the guidance needed to implement activities, but the plan cannot be static. Monitoring the performance of your management actions is essential to understanding whether your goals have been met, and whether further actions are needed. Monitoring and evaluating the implementation and effectiveness of a lake watershed management plan allows assessment of progress towards the goals and objectives of the plan, identification of problems and opportunities, and a collection of critical information required when performing a 5 or 10 year review of the plan.
The development of a lake watershed management plan provides the guidance needed to implement activities, but the plan cannot be static. Monitoring the performance of your management actions is essential to understanding whether your goals have been met, and whether further actions are needed. Monitoring and evaluating the implementation and effectiveness of a lake watershed management plan allows assessment of progress towards the goals and objectives of the plan, identification of problems and opportunities, and a collection of critical information required when performing a 5 or 10 year review of the plan.
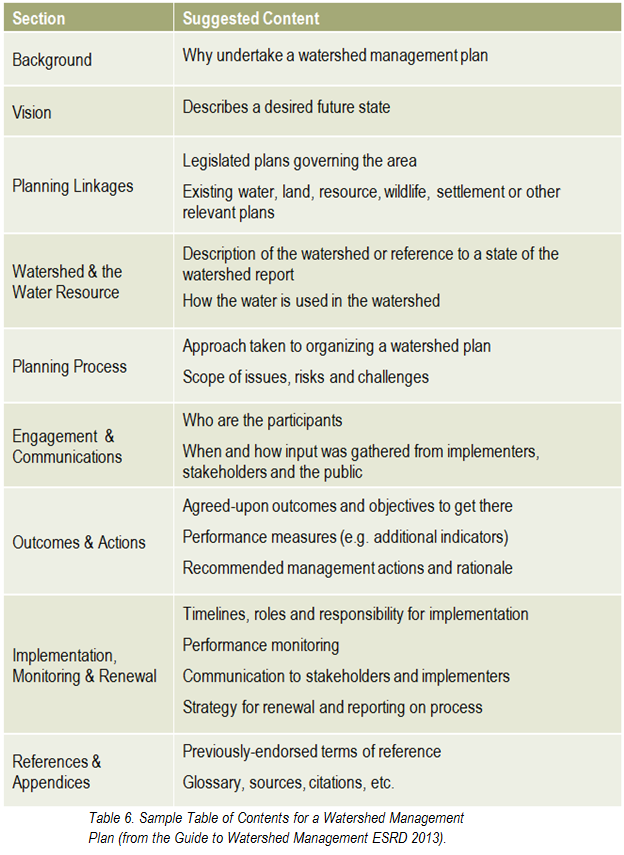
 What has the monitoring results of the plan and of the indicators shown? Is there a need to modify the plan? It is important that the lake watershed management plan does not just sit on a shelf. Information gaps should be addressed, action items need to be managed, completed, and evaluated to best address the needs of the lake. Always keep in mind the vision: if the actions taken are not bringing the lake closer to that vision, then the plan needs to be modified. Consider updating both the state of the watershed and the lake watershed management plans at regular intervals to make sure that the actions taken were achieving the desired outcomes and to evaluate what work still needs to be done.
What has the monitoring results of the plan and of the indicators shown? Is there a need to modify the plan? It is important that the lake watershed management plan does not just sit on a shelf. Information gaps should be addressed, action items need to be managed, completed, and evaluated to best address the needs of the lake. Always keep in mind the vision: if the actions taken are not bringing the lake closer to that vision, then the plan needs to be modified. Consider updating both the state of the watershed and the lake watershed management plans at regular intervals to make sure that the actions taken were achieving the desired outcomes and to evaluate what work still needs to be done. Once a plan has been approved by all affected sectors and officially endorsed and released by the steering committee, then implementation can begin in full. Action projects can be large and comprehensive, or made smaller by staging projects over time or into modules that can be tackled one at a time. Fundraising is an issue that many community groups may find intimidating, but experience with programs such as the Pine Lake Restoration Program (see
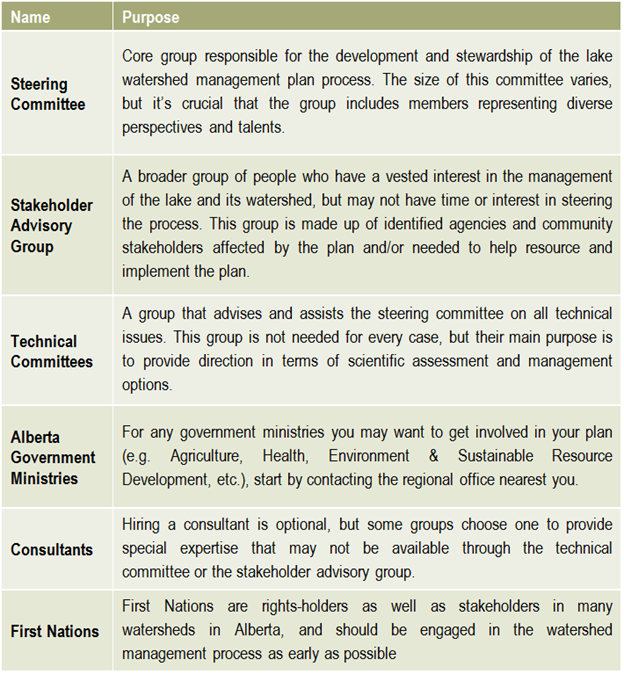
Once a plan has been approved by all affected sectors and officially endorsed and released by the steering committee, then implementation can begin in full. Action projects can be large and comprehensive, or made smaller by staging projects over time or into modules that can be tackled one at a time. Fundraising is an issue that many community groups may find intimidating, but experience with programs such as the Pine Lake Restoration Program (see 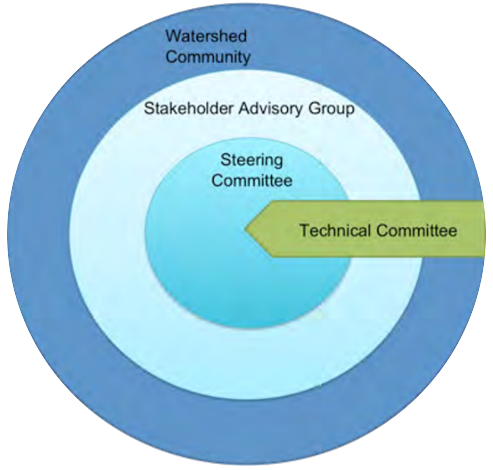 This graphic describes how the various committees and groups will work and interact together. The circle size depicts the approximate number of people involved, and the circles overlapping indicates that some individuals may reside in all of the circles and participate in multiple committees as part of the planning process. The technical committee is shown as an arrow, indicating that it is independent and has relatively few people, and yet it interacts with all of the groups. This graphic may look different depending on the lake and the people involved, and a detailed structure should be agreed upon and described in the plan’s Terms of Reference (Step 6).
This graphic describes how the various committees and groups will work and interact together. The circle size depicts the approximate number of people involved, and the circles overlapping indicates that some individuals may reside in all of the circles and participate in multiple committees as part of the planning process. The technical committee is shown as an arrow, indicating that it is independent and has relatively few people, and yet it interacts with all of the groups. This graphic may look different depending on the lake and the people involved, and a detailed structure should be agreed upon and described in the plan’s Terms of Reference (Step 6).